Description
| Assay type: | Sandwich |
| Format: | 96T |
| Assay time: | 4.5h |
| Reactivity: | Rat |
| Detection method: | Chemiluminescence |
| Detection range: | 12.50-800 pg/mL |
| Sensitivity: | 7.50 pg/mL |
| Sample volume: | 100µL |
| Sample type: | Serum, plasma and other biological fluids |
| Repeatability: | CV < 15% |
| Specificity: | This kit recognizes Rat LEPR in samples. No significant cross-reactivity or interference between Rat LEPR and analogues was observed. |
This kit uses Sandwich-CLIA as the method. The micro CLIA plate provided in this kit has been pre-coated with an antibody specific to Rat LEPR. Standards or samples are added to the appropriate micro CLIA plate wells and combined with the specific antibody. Then a biotinylated detection antibody specific for Rat LEPR and Avidin-Horseradish Peroxidase (HRP) conjugate are added to each micro plate well successively and incubated. Free components are washed away. The substrate solution is added to each well. Only those wells that contain Rat LEPR, biotinylated detection antibody and Avidin-HRP conjugate will appear fluorescence. The Relative light unit (RLU) value is measured spectrophotometrically by the Chemiluminescence immunoassay analyzer. The RLU value is positively associated with the concentration of Rat LEPR. The concentration of Rat LEPR in the samples can be calculated by comparing the RLU of the samples to the standard curve.
| UniProt Protein Function: | Receptor for hormone LEP/leptin (Probable). On ligand binding, mediates LEP central and peripheral effects through the activation of different signaling pathways such as JAK2/STAT3 and MAPK cascade/FOS. In the hypothalamus, LEP acts as an appetite-regulating factor that induces a decrease in food intake and an increase in energy consumption by inducing anorexinogenic factors and suppressing orexigenic neuropeptides, also regulates bone mass and secretion of hypothalamo-pituitary-adrenal hormones (PubMed:8690163). In the periphery, increases basal metabolism, influences reproductive function, regulates pancreatic beta-cell function and insulin secretion, is pro-angiogenic and affects innate and adaptive immunity. Control of energy homeostasis and melanocortin production (stimulation of POMC and full repression of AgRP transcription) is mediated by STAT3 signaling, whereas distinct signals regulate NPY and the control of fertility, growth and glucose homeostasis. Involved in the regulation of counter-regulatory response to hypoglycemia by inhibiting neurons of the parabrachial nucleus. Has a specific effect on T lymphocyte responses, differentially regulating the proliferation of naive and memory T-cells. Leptin increases Th1 and suppresses Th2 cytokine production. |
| NCBI Summary: | receptor for leptin; may have a role in body weight maintainence [RGD, Feb 2006] |
| UniProt Code: | Q62959 |
| NCBI GenInfo Identifier: | 6981150 |
| NCBI Gene ID: | 24536 |
| NCBI Accession: | NP_036728. 1 |
| UniProt Secondary Accession: | Q62959,O35772, O35773, O54805, P70493, P70494, P70495 P97589, Q62960, Q63007, Q63385, Q63386, |
| UniProt Related Accession: | Q62959 |
| Molecular Weight: | 101,395 Da |
| NCBI Full Name: | leptin receptor |
| NCBI Synonym Full Names: | leptin receptor |
| NCBI Official Symbol: | Lepr |
| NCBI Official Synonym Symbols: | Fa |
| NCBI Protein Information: | leptin receptor |
| UniProt Protein Name: | Leptin receptor |
| UniProt Synonym Protein Names: | OB receptor; OB-R; CD_antigen: CD295 |
| Protein Family: | Leptin receptor |
| UniProt Gene Name: | Lepr |
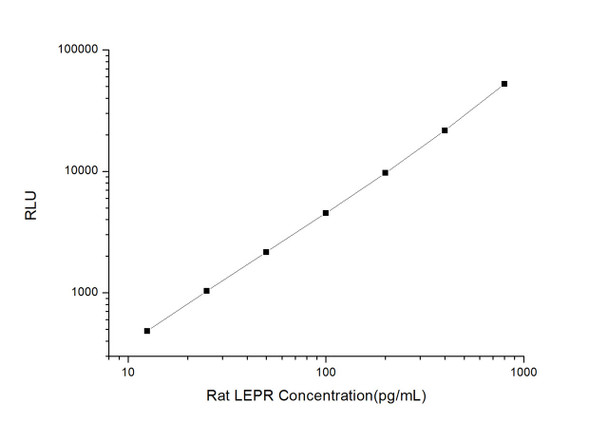
As the RLU values of the standard curve may vary according to the conditions of the actual assay performance (e. g. operator, pipetting technique, washing technique or temperature effects), the operator should establish a standard curve for each test. Typical standard curve and data is provided below for reference only.
| Concentration (pg/mL) | RLU | Average | Corrected |
| 800 | 50226 55058 | 52642 | 52616 |
| 400 | 21453 22015 | 21734 | 21708 |
| 200 | 9851 9565 | 9708 | 9682 |
| 100 | 4286 4818 | 4552 | 4526 |
| 50 | 2233 2143 | 2188 | 2162 |
| 25 | 1121 999 | 1060 | 1034 |
| 12.50 | 490 528 | 509 | 483 |
| 0 | 25 27 | 26 | -- |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, mid range and high level Rat LEPR were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, mid range and high level Rat LEPR were tested on 3 different plates, 20 replicates in each plate.
| Intra-assay Precision | Inter-assay Precision | |||||
| Sample | 1 | 2 | 3 | 1 | 2 | 3 |
| n | 20 | 20 | 20 | 20 | 20 | 20 |
| Mean (pg/mL) | 36.78 | 78.59 | 296.14 | 39.21 | 78.46 | 286.07 |
| Standard deviation | 4.68 | 7.76 | 29.79 | 3.18 | 7.01 | 20.31 |
| C V (%) | 12.72 | 9.87 | 10.06 | 8.11 | 8.93 | 7.10 |
Recovery
The recovery of Rat LEPR spiked at three different levels in samples throughout the range of the assay was evaluated in various matrices.
| Sample Type | Range (%) | Average Recovery (%) |
| Serum (n=5) | 92-106 | 98 |
| EDTA plasma (n=5) | 84-97 | 91 |
| Cell culture media (n=5) | 89-101 | 95 |
Linearity
Samples were spiked with high concentrations of Rat LEPR and diluted with Reference Standard & Sample Diluent to produce samples with values within the range of the assay.
| Serum (n=5) | EDTA plasma (n=5) | Cell culture media (n=5) | ||
| 1:2 | Range (%) | 101-114 | 87-101 | 86-101 |
| Average (%) | 107 | 93 | 93 | |
| 1:4 | Range (%) | 103-119 | 85-97 | 90-102 |
| Average (%) | 110 | 92 | 95 | |
| 1:8 | Range (%) | 90-104 | 85-97 | 96-112 |
| Average (%) | 95 | 92 | 103 | |
| 1:16 | Range (%) | 96-112 | 100-115 | 89-101 |
| Average (%) | 103 | 108 | 95 |
An unopened kit can be stored at 4°C for 1 month. If the kit is not used within 1 month, store the items separately according to the following conditions once the kit is received.
| Item | Specifications | Storage |
| Micro CLIA Plate(Dismountable) | 8 wells ×12 strips | -20°C, 6 months |
| Reference Standard | 2 vials | |
| Concentrated Biotinylated Detection Ab (100×) | 1 vial, 120 µL | |
| Concentrated HRP Conjugate (100×) | 1 vial, 120 µL | -20°C(shading light), 6 months |
| Reference Standard & Sample Diluent | 1 vial, 20 mL | 4°C, 6 months |
| Biotinylated Detection Ab Diluent | 1 vial, 14 mL | |
| HRP Conjugate Diluent | 1 vial, 14 mL | |
| Concentrated Wash Buffer (25×) | 1 vial, 30 mL | |
| Substrate Reagent A | 1 vial, 5 mL | 4°C (shading light) |
| Substrate Reagent B | 1 vial, 5 mL | 4°C (shading light) |
| Plate Sealer | 5 pieces | |
| Product Description | 1 copy | |
| Certificate of Analysis | 1 copy |
- Set standard, test sample and control (zero) wells on the pre-coated plate and record theirpositions. It is recommended to measure each standard and sample in duplicate. Note: addall solutions to the bottom of the plate wells while avoiding contact with the well walls. Ensuresolutions do not foam when adding to the wells.
- Aliquot 100µl of standard solutions into the standard wells.
- Add 100µl of Sample / Standard dilution buffer into the control (zero) well.
- Add 100µl of properly diluted sample (serum, plasma, tissue homogenates and otherbiological fluids. ) into test sample wells.
- Cover the plate with the sealer provided in the kit and incubate for 90 min at 37°C.
- Aspirate the liquid from each well, do not wash. Immediately add 100µL of BiotinylatedDetection Ab working solution to each well. Cover the plate with a plate seal and gently mix. Incubate for 1 hour at 37°C.
- Aspirate or decant the solution from the plate and add 350µL of wash buffer to each welland incubate for 1-2 minutes at room temperature. Aspirate the solution from each well andclap the plate on absorbent filter paper to dry. Repeat this process 3 times. Note: a microplatewasher can be used in this step and other wash steps.
- Add 100µL of HRP Conjugate working solution to each well. Cover with a plate seal andincubate for 30 min at 37°C.
- Aspirate or decant the solution from each well. Repeat the wash process for five times asconducted in step 7.
- Add 100µL of Substrate mixture solution to each well. Cover with a new plate seal andincubate for no more than 5 min at 37°C. Protect the plate from light.
- Determine the RLU value of each well immediately.
Additional Information
Product Type: |
ELISA Kit |
ELISA Type: |
CLIA Kit |
Size: |
96 Assays |
Sensitivity: |
7.5pg/mL |
Range: |
12.5-800pg/mL |
ELISA Type: |
Sandwich |
Synonyms: |
CD295, LEP-R, LR, LEPRD, OB-R, OBR |
Reactivity: |
Rat |
Sample Type: |
Serum, plasma and other biological fluids |