Cell Cycle Recombinant Proteins
Human PIN1/Rotamase Pin1 Recombinant Protein (RPES4818)
- SKU:
- RPES4818
Description
Human PIN1/Rotamase Pin1 Recombinant Protein
Peptidyl-prolyl cis-trans isomerase Pin1; also known as Peptidyl-prolyl cis-trans isomerase NIMA-interacting 1; Rotamase Pin1 and PIN1; peptidyl-prolyl cis/trans isomerase (PPIase); is a nucleus protein. PIN1 is a peptidyl-prolyl isomerase that can alter the conformation of phosphoproteins and so affect protein function and/or stability. PIN1 regulates a number of proteins important for cell-cycle progression and is presumed to operate as a molecular timer of this important process. PIN1 is an essential PPIase that regulates mitosis presumably by interacting with NIMA and attenuating its mitosis-promoting activity. PIN1 displays a preference for an acidic residue N-terminal to the isomerized proline bond. Alterations in the level of PIN1 can influence hyperproliferative diseases such as cancer. PIN1 has been implicated in multiple aspects of cell cycle regulation. It has been suggested that PIN1 function is required for both normal mitotic progression and reentry into the cell cycle from quiescence. PIN1 is also a target of several oncogenic pathways and is overexpressed in human breast cancer. Its overexpression can lead to upregulation of cyclin-D1 and transformation of breast epithelial cells in collaboration with the oncogenic pathways. PIN1 plays a pivotal role in breast development and may be a promising new anticancer target. Pin1 activity regulates the outcome of proline-directed kinase (e.g. MAPK; CDK or GSK3) signalling and consequently regulates cell proliferation (in part through control of cyclin D1 levels and stability) and cell survival. Recent data also implicate Pin1 as playing an important role in immune responses; at least in part by increasing the stability of cytokine mRNAs by influencing the protein complexes to which they bind.
| Product Name: | Human PIN1/Rotamase Pin1 Recombinant Protein (RPES4818) |
| Product Code: | RPES4818 |
| Size: | 50µg |
| Species: | Human |
| Expressed Host: | E.coli |
| Synonyms: | DOD,UBL5 |
| Accession: | Q13526-1 |
| Sequence: | Met 1-Glu 163 |
| Fusion tag: | N-His |
| Endotoxin: | Please contact us for more information. |
| Protein Construction: | A DNA sequence encoding the mature form of human PIN1 (Q13526-1) (Met 1-Glu 163) was expressed; with a polyhistide tag at the N-terminus. |
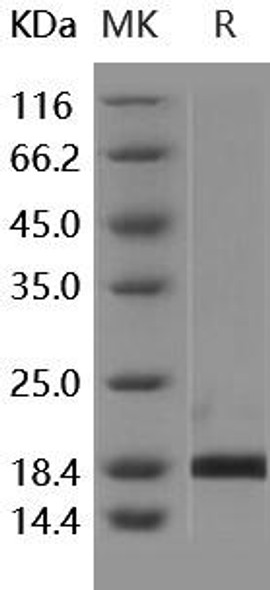
| Purity: | > 90 % as determined by reducing SDS-PAGE. |
| Mol Mass: | 20.3 kDa |
| AP Mol Mass: | 21 kDa |
| Formulation: | Lyophilized from sterile PBS; pH 7.4 |
| Shipping: | This product is provided as lyophilized powder which is shipped with ice packs. |
| Stability and Storage: | Lyophilized proteins are stable for up to 12 months when stored at -20 to -80°C. Reconstituted protein solution can be stored at 4-8°C for 2-7 days. Aliquots of reconstituted samples are stable at < -20°C for 3 months. |
Additional Information
Product Type: |
Recombinant Protein |
Species: |
Human |
Research Area: |
Cell Cycle |