Description
| Antibody Name: | NARF Antibody (PACO51738) |
| Antibody SKU: | PACO51738 |
| Size: | 50ug |
| Host Species: | Rabbit |
| Tested Applications: | ELISA, WB, IF |
| Recommended Dilutions: | ELISA:1:2000-1:10000, WB:1:500-1:5000, IF:1:50-1:200 |
| Species Reactivity: | Human, Mouse |
| Immunogen: | Recombinant Human Nuclear prelamin A recognition factor protein (10-41AA) |
| Form: | Liquid |
| Storage Buffer: | Preservative: 0.03% Proclin 300 Constituents: 50% Glycerol, 0.01M PBS, pH 7.4 |
| Purification Method: | >95%, Protein G purified |
| Clonality: | Polyclonal |
| Isotype: | IgG |
| Conjugate: | Non-conjugated |
 | Western Blot. Positive WB detected in: A549 whole cell lysate, MCF-7 whole cell lysate, Mouse kidney tissue, Mouse skeletal muscle tissue. All lanes: NARF antibody at 3µg/ml. Secondary. Goat polyclonal to rabbit IgG at 1/50000 dilution. Predicted band size: 52, 57, 46, 45 kDa. Observed band size: 52 kDa.. |
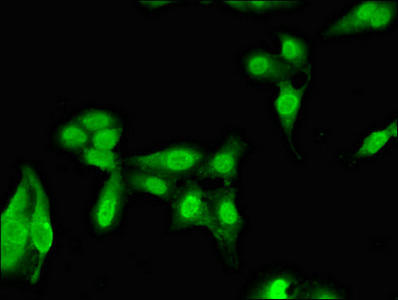 | Immunofluorescent analysis of Hela cells using PACO51738 at dilution of 1:100 and Alexa Fluor 488-congugated AffiniPure Goat Anti-Rabbit IgG(H+L). |
| Background: | lamin filament, nuclear lamina, nuclear lumen, lamin binding |
| Synonyms: | Nuclear prelamin A recognition factor (Iron-only hydrogenase-like protein 2) (IOP2), NARF |
| UniProt Protein Function: | NARF: Several proteins have been found to be prenylated and methylated at their carboxyl-terminal ends. Prenylation was initially believed to be important only for membrane attachment. However, another role for prenylation appears to be its importance in protein-protein interactions. The only nuclear proteins known to be prenylated in mammalian cells are prelamin A- and B-type lamins. Prelamin A is farnesylated and carboxymethylated on the cysteine residue of a carboxyl-terminal CaaX motif. This post-translationally modified cysteine residue is removed from prelamin A when it is endoproteolytically processed into mature lamin A. The protein encoded by this gene binds to the prenylated prelamin A carboxyl-terminal tail domain. It may be a component of a prelamin A endoprotease complex. The encoded protein is located in the nucleus, where it partially colocalizes with the nuclear lamina. It shares limited sequence similarity with iron-only bacterial hydrogenases. Alternatively spliced transcript variants encoding different isoforms have been identified for this gene, including one with a novel exon that is generated by RNA editing. [provided by RefSeq, Jul 2008]Chromosomal Location of Human Ortholog: 17q25.3Cellular Component: lamin filament; nuclear lamina; nuclear lumenMolecular Function: lamin binding |
| UniProt Protein Details: | |
| NCBI Summary: | Several proteins have been found to be prenylated and methylated at their carboxyl-terminal ends. Prenylation was initially believed to be important only for membrane attachment. However, another role for prenylation appears to be its importance in protein-protein interactions. The only nuclear proteins known to be prenylated in mammalian cells are prelamin A- and B-type lamins. Prelamin A is farnesylated and carboxymethylated on the cysteine residue of a carboxyl-terminal CaaX motif. This post-translationally modified cysteine residue is removed from prelamin A when it is endoproteolytically processed into mature lamin A. The protein encoded by this gene binds to the prenylated prelamin A carboxyl-terminal tail domain. It may be a component of a prelamin A endoprotease complex. The encoded protein is located in the nucleus, where it partially colocalizes with the nuclear lamina. It shares limited sequence similarity with iron-only bacterial hydrogenases. Alternatively spliced transcript variants encoding different isoforms have been identified for this gene, including one with a novel exon that is generated by RNA editing. [provided by RefSeq, Jul 2008] |
| UniProt Code: | Q9UHQ1 |
| NCBI GenInfo Identifier: | 74735021 |
| NCBI Gene ID: | 26502 |
| NCBI Accession: | Q9UHQ1.1 |
| UniProt Secondary Accession: | Q9UHQ1,Q96AY9, Q9BWC6, A6NCJ3, B3KPX2, K4DI98 |
| UniProt Related Accession: | Q9UHQ1 |
| Molecular Weight: | 44,681 Da |
| NCBI Full Name: | Nuclear prelamin A recognition factor |
| NCBI Synonym Full Names: | nuclear prelamin A recognition factor |
| NCBI Official Symbol: | NARF |
| NCBI Official Synonym Symbols: | IOP2 |
| NCBI Protein Information: | nuclear prelamin A recognition factor |
| UniProt Protein Name: | Nuclear prelamin A recognition factor |
| UniProt Synonym Protein Names: | Iron-only hydrogenase-like protein 2; IOP2 |
| Protein Family: | Nuclear prelamin A recognition factor |
| UniProt Gene Name: | NARF |
| UniProt Entry Name: | NARF_HUMAN |
Additional Information
Product Type: |
Antibody |
Reactivity: |
Human |
Reactivity: |
Mouse |
Host Species: |
Rabbit |
Isotype: |
IgG |
Applications: |
ELISA |
Applications: |
WB |
Applications: |
IF |
Antibody Type: |
Polyclonal Antibody |
Conjugation: |
Unconjugated |






