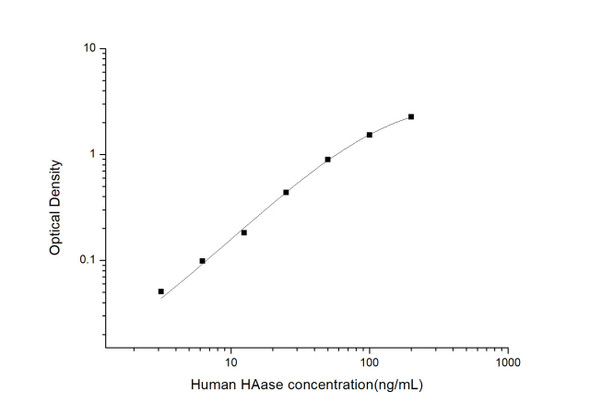
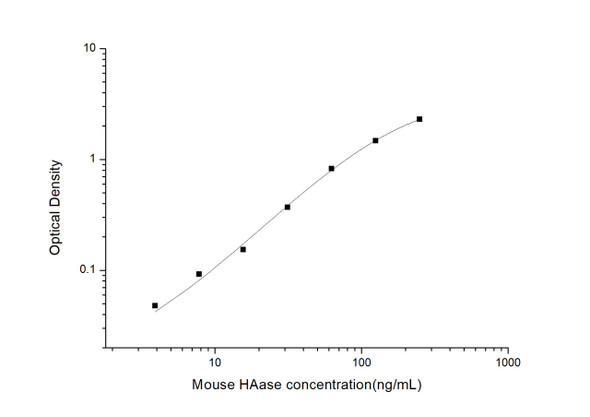
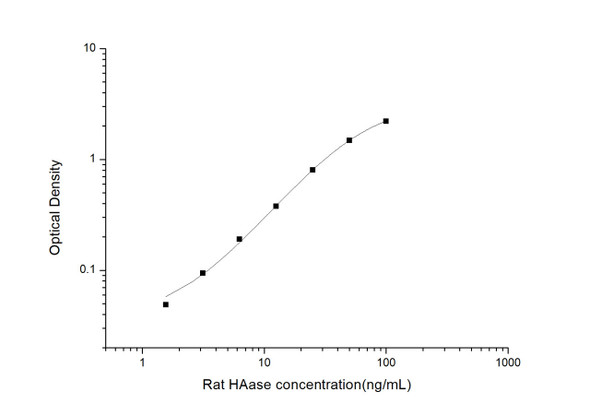
Description
system_update_altDatasheet
| Product Name: | Human Hyaluronidase (HAase) ELISA Kit |
| Product Code: | HUDL01295 |
| Size: | 96 Assays |
| Target: | HAase |
| Synonyms: | Hyaluronic Lase, Hyaluroniclase |
| Detection Method: | Sandwich |
| Range: | 3.125-200ng/mL |
| Shelf Life: | 12 months |
| Note: | For research use only |
| Recovery: | Matrices listed below were spiked with certain level of recombinant the index and the recovery rates were calculated by comparing the measured value to the expected amount of the index in samples. | ||||||||||||||||||||
| |||||||||||||||||||||
| Linearity: | The linearity of the kit was assayed by testing samples spiked with appropriate concentration of the index and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected. | ||||||||||||||||||||
| |||||||||||||||||||||
| Intra-assay Precision: | Intra-Assay: CV <10%. 3 samples with low, middle and high level the index were tested 20 times on one plate, respectively. | ||||||||||||||||||||
| Inter-assay Precision: | Inter-Assay: CV <12%. 3 samples with low, middle and high level the index were tested on 3 different plates, 8 replicates in each plate. | ||||||||||||||||||||
| Stability: | The stability of ELISA kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage conditions. Note: To minimize unnecessary influences on the performance, operation procedures and lab conditions, especially room temperature, air humidity and incubator temperatures should be strictly regulated. It is also strongly suggested that the whole assay is performed by the same experimenter from the beginning to the end. |
| Step | Protocol |
| 1. | Prepare all reagents, samples and standards |
| 2. | Add 100µL standard or sample to each well. Incubate 2 hours at 37°C |
| 3. | Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C |
| 4. | Aspirate and wash 3 times |
| 5. | Add 100µL prepared Detection Reagent B. Incubate 1 hour at 37°C |
| 6. | Aspirate and wash 5 times |
| 7. | Add 90µL Substrate Solution. Incubate 15-25 minutes at 37°C |
| 8. | Add 50µL Stop Solution. Read at 450nm immediately. |
Additional Information
Product Type: |
ELISA Kit |
Sensitivity: |
1.28ng/mL |
Range: |
3.125-200ng/mL |
ELISA Type: |
Sandwich |
Synonyms: |
Hyaluronic Lase,Hyaluroniclase |
Reactivity: |
Human |
Sample Type: |
Serum, Plasma, Tissue Homogenates And Other Biological Fluids |